Part 1 will focus on monstrosities involving various combinations of beak and eye anomalies, in limbs and trunk, classical celosomia and some deformities involving the embryo’s brain and spinal cord.
...
-->Introduction
-->Malformations of Limbs and Trunk
--> Deformities in the Embryo’s brain
...
TRENDS IN DEVELOPMENTAL ANOMALIES IN CONTEMPORARY BROILER CHICKENS
By Dr. Andrew A. Olkowski, DVM and Dr. Bernard Laarveld, Department of Animal and Poultry Science, and Chris Wojnarowicz, MVSc, DVM, Prairie Diagnostic Services, Department of Veterinary Pathology, University of Saskatchewan, Saskatoon, SK S7N 5A8, Canada and Cindy Pentelichuk, QA Manager, Lilydale Wynyard Hatchery, Sofina Foods Company, 605 Greer St, Wynyard, SK S0A 4T0, Canada.
.
Introduction
For many years the broiler industry has focused on genetic selection for rapid gain of muscle mass, decreased time from hatch to market, and increased feed efficiency. This strategy has resulted in a chicken genotype with superior growth characteristics.
However, in association with the successful development of economically important targets, some undesirable traits have become apparent. In many ways, experience from the work on genetic improvement of broilers has provided a unique insight into understanding the effects of ‘human controlled evolution’ on overall health.
.
Variety of Health Problems
Over the 60+ years’ history of intensive selection, the broiler industry has witnessed a variety of health problems involving muscular, digestive, cardiovascular, integumentary, skeletal, and immune systems. Many debilitating diseases marring modern broiler production are well documented, but this represents only one aspect of the health problems.
Recently completed sections of our long term study revealed trends in the incidence of anatomical anomalies in contemporary broilers.
The occurrence of embryonic abnormalities signifies a discreet category of health problems, and careful monitoring of congenital anomalies provides very valuable information regarding the overall health status of the population.
Nevertheless, with the exception of a handful of incidental reports, the research on embryonic anomalies in broilers has been dormant during the last several decades. Moreover, there is a dearth of epidemiological information on naturally occurring anomalies in modern commercial broilers.
Therefore, a research project was developed to examine both type and incidence of anatomical anomalies occurring in commercial broilers.
The study was conducted at the University of Saskatchewan, Canada in collaboration with Lilydale Wynyard Hatchery, Sofina Foods Company.
^ Top page
.
Extensive Data
The data gathering activities commenced in 2003 and included examination of eggs that failed to hatch, newly hatched chicks, and monitoring of commercial flocks for the occurrence of anatomical anomalies. The study is ongoing, and we plan to continue this activity into the foreseeable future.
During the course of our research, we documented practically all types of monstrosities reported previously in avian species. However, we also documented several embryonic defects that were previously not observed in broilers.
^ Top page
.
Beak and Eye Anomalies
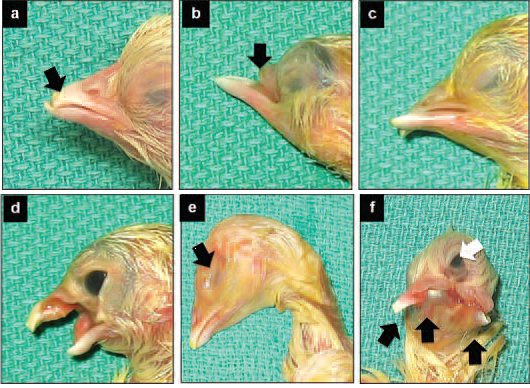
All common anomalies were observed in all strains of broilers used for commercial purposes in the province of Saskatchewan. Among the more common deformities observed in our study were anomalies involving beak and eyes (Figure 1).
Figure 1 Monstrosities involving various combinations of beak and eye anomalies.
Common beak anomalies included underdeveloped upper beak (Figure 1a, arrow), lack of upper beak (Figure 1b, arrow) and cross beak (Figure 1c). Occasionally, deformity characterised by growth of soft tissue inside of the mouth (Figure 1d) was observed.
Common anomalies of eyes included lack of eyes (anopthalmia) shown in Figure 1e (arrow), and single eye (cyclopia) shown in Figure 1f (white arrow). Noteworthy are multiple monstrosities in Figure 1f showing in addition to cyclopia, multiple beaks (black arrows).
^ Top page
.
Malformations of Limbs and Trunk
Various malformations of limbs and trunk were also a common finding. Anomalies of both wings and legs were observed, but in the majority of cases deformities were predominantly confined to legs.
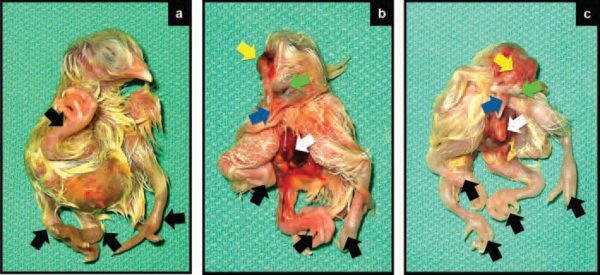
The affected embryos showed grossly disfigured legs (mostly at joint levels) and frequently presented extra members or multiple sets of legs, but more typically limb deformities occurred together with several other anomalies (Figure 2).
Figure2 Monstrosities primarily affecting the trunk of the body presented as sole leg deformity, or multiple monstrosities involving legs, cranium, beak, and viscera. Figure 2a shows a near term embryo with four legs (arrow). This monstrosity is not necessarily lethal, and some chicks with this anomaly may hatch successfully. In many instances, embryos with leg deformities (Figure 2b and 2c, black arrows) also showed multiple anomalies including exposed brain (yellow arrows), eyes anomalies (green arrows), beak deformities (blue arrows) and exposed internal organs due to failure of abdominal closure (Figure 2b and 2c, white arrows). Monstrosities shown in Figure 2b and 2c are lethal.
Very common deformities encountered in our study can generally be characterised as protrusion of the viscera through an open abdominal wall in otherwise normally developing embryo (Figure 3).
^ Top page
.
Classical Celosomia
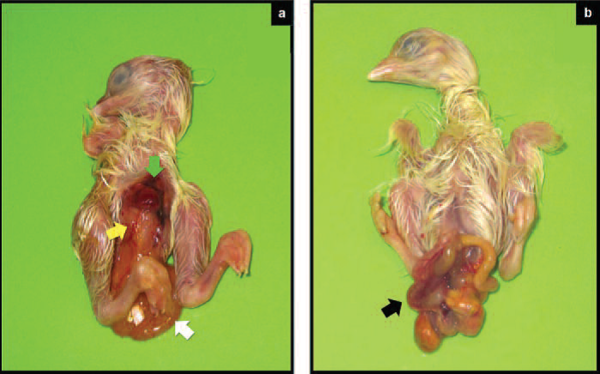
Figure 3 Exposed viscera in 18-20 day old broiler chicken embryos. The landmark of this embryonic pathology includes exposed viscera due to failure to close coelomic cavity as presented in Figure 3a, where failure to close coelomic cavity resulted in total exposure of internal organs including heart (green arrow), liver (yellow arrow), and gastrointestinal tract (white arrow). In addition to this, we also observed many cases with partially incomplete closure of abdominal wall as presented in Figure 3b (arrow).
In the older literature, the monstrosity shown in Figure 3a was described as celosomia attributed to the underdeveloped sternal crest and caudo-lateral processes, associated with autosomal recessive gene.
In the present study, we observed many cases consistent with the classical form of celosomia where all internal organs (heart, liver, and entire gastrointestinal tract) are exposed.
However, we also noted another anomaly characterised by partial incomplete closure of the abdominal wall where only some segments of intestines were exposed (Figure 3b, black arrow). In many cases, the intestines are incarcerated by the closing abdominal muscle, which resulted in severe congestion and necrosis.
This form of anatomical anomaly has not been reported previously in commercial broilers, and was described in detail only recently (Wojnarowicz and Olkowski, Avian Pathology, 2009, 38:509-512). Since our first report, the incidence of this anomaly has shown increasing trends.
^ Top page
.
Deformities in the Embryo’s brain
The common (and probably most intriguing) anomalies seen in our study were in the general category of deformities involving the embryo’s brain and spinal cord.
Representative examples of various forms of these anomalies are shown in Figure 4.
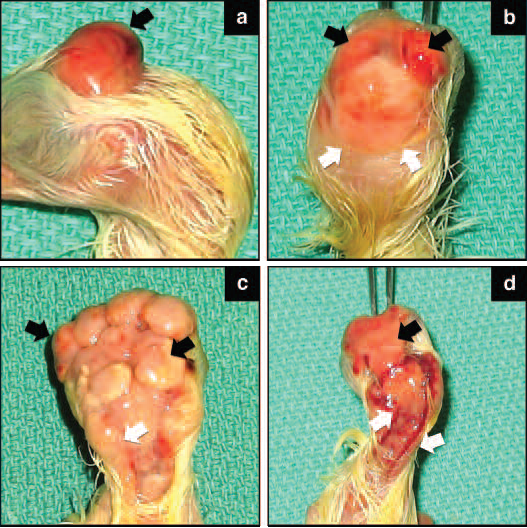
Figure 4 Typical brain and spinal cord anomalies seen in 18-20 day old broiler embryos.
The commonly observed brain anomalies were characterised as partial absence of some components of the cranial bones (Figure 4a, arrow), or total absence of the parietal, occipital and temporal bones, with the entire brain being fully exposed (Figure 4b, arrows). In some cases brain anomalies involved structures resembling multiple brains (Figure 4c, black arrows). Many embryos showed anomalies of the spinal cord known as spina bifida (Figure 4d, white arrows).
Deformities such as open cerebral cavity (acrania) with brain structures exposed were previously reported. However, the occurrence of anomalies such as excessive brain structures or deformity of the spinal cord has not been observed in commercial broilers prior to discovery in our studies. The pathology of Spina bifida in commercial broilers was described in detail only recently.
^ Top page
This article appeared in International Hatchery Practice, Volume 28 Number 1 (2013). ©Copyright 2013, All Rights Reserved.
Part 2 of this article on ‘Trends in developmental anomalies in contemporary broiler chickens’ was featured in International Hatchery Practice, Volume 28 Number 2 (2013).
<< Back to page Other Informations
Next to Part 2 - Trend in developmental anomalies in contemporary broiler chickens ≫

 Corporate Website
Corporate Website
 Africa
Africa
 Argentina
Argentina
 Asia
Asia
 Australia
Australia
 Belgium
Belgium
 Brazil
Brazil
 Bulgaria
Bulgaria
 Canada (EN)
Canada (EN)
 Chile
Chile
 China
China
 Colombia
Colombia
 Denmark
Denmark
 Egypt
Egypt
 France
France
 Germany
Germany
 Greece
Greece
 Hungary
Hungary
 Indonesia
Indonesia
 Italia
Italia
 India
India
 Japan
Japan
 Korea
Korea
 Malaysia
Malaysia
 Mexico
Mexico
 Middle East
Middle East
 Netherlands
Netherlands
 Peru
Peru
 Philippines
Philippines
 Poland
Poland
 Portugal
Portugal
 Romania
Romania
 Russia
Russia
 South Africa
South Africa
 Spain
Spain
 Sweden
Sweden
 Thailand
Thailand
 Tunisia
Tunisia
 Turkey
Turkey
 Ukraine
Ukraine
 United Kingdom
United Kingdom
 USA
USA
 Vietnam
Vietnam