...
--> Introduction
--> Molecular diagnostic methods
--> Vector vaccines
...
MANAGEMENT OF RESPIRATORY DISEASES IN POULTRY
By Kenton Kreager, DVM, Hy-ILine International, Dallas Center, Iowa, USA
.
INTRODUCTION
Many of the most common and serious infectious diseases in layers are respiratory diseases. These include the bacterial diseases Mycoplasma gallisepticum (MG) and Infectious Coryza (Avibacterium paragallinarum), and viral diseases such as Newcastle disease, Infectious Bronchitis, Infectious Laryngotracheitis, Avian Influenza, and Avian Metapneumovirus.
Each disease has its unique characteristics regarding the nature of the causative organism, avian species affected, transmission routes, and control or vaccination options.
While these diseases can often appear quite similar clinically and are difficult to distinguish based only on symptoms and lesions, it is necessary to accurately diagnose them and consider each as a separate and unique disease.
Clinical symptoms of respiratory disease are seldom diagnostic of a specific disease. Combinations of these respiratory diseases can occur within the same flock, further complicating the diagnosis of the causative agents and making the clinical situation worse. Often one of the bacterial diseases (MG or Coryza) may be already present in a flock in a chronic form, and is then exacerbated by an overriding infection of a virus like infectious bronchitis or Newcastle disease. The combined effect of both diseases together is much more severe and long lasting than either would be alone, and is typically then referred to as Chronic Respiratory Disease (CRD).
It is easy to observe the clinical effects of respiratory disease and tempting to treat it all the same with broad spectrum antibiotics, but in order to get good control, it is necessary to use proper diagnostic methods to accurately identify the individual diseases involved and design appropriate future control strategies for each.
Postmortem examinations, histopathology, bacterial culture / identification, serology, and molecular methods (PCR) can all be useful in identifying the various causes of respiratory diseases.
Image 1: Each disease needs to have good diagnostics to determine what the problems are so they can be addressed individually
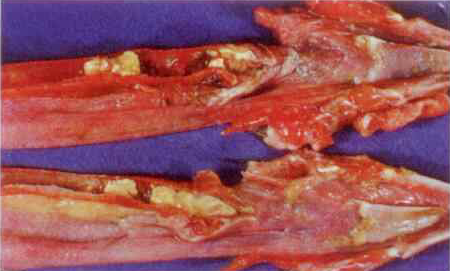
Postmortem examinations alone may be diagnostic for some diseases, like parasitism or enteritis, but most respiratory diseases look similar enough that further diagnostic methods need to be used to distinguish them. Histopathology done on formalin-fixed tissues can reveal cellular lesions that are diagnostic for fowl pox (wet or dry forms) and infectious laryngotracheitis.
Wet pox in the trachea can appear very similar to LT, so histopathology should always be done when either disease is suspected. Other than that, histopathology may demonstrate general respiratory lesions, but is not diagnostic for specifically identifying the other respiratory diseases.
Image 2: Severe tracheitis - LT or pox?
.
MOLECULAR DIAGNOSTIC METHODS
While isolating and identifying the causative organism is the most positive method of diagnosis, it often is not practical. Some pathogenic bacteria are relatively easy to grow in a laboratory, like E. coli, or Salmonella, but the bacterial respiratory diseases, MG and Coryza, are caused by bacteria that require special growth media and incubation conditions.
Viruses are also relatively difficult to isolate and identify as they need other living cells to reproduce in, such as embryonated chicken eggs or a tissue culture substrate. For diagnostic purposes, newer molecular diagnostic methods, such as PCR, have replaced the need for actually growing and identifying some of these pathogenic micro-organisms.
PCR identifies the presence of a gene or segment of DNA that is unique to a particular organism. It is very sensitive and specific and, as such, is equal in value to isolating and identifying the organism. Commercial PCR systems have made this technology available for many diagnostic laboratories.
Biosecurity programs should be designed to prevent a farm from becoming infected with new diseases it does not already have. The specific characteristics of those diseases should be considered regarding potential sources of the disease agent, transmissibility, and survival in the environment.
Common biosecurity risks can come from other animal species on the farm (domesticated or wild), people who may have had other poultry contact, airborne transmission from other farms or houses, portable equipment that has been on other farms, new flocks being moved onto the farm, and the consumed items, water and feed.
Farm managers need to consider the biosecurity risks most important for them and design prevention programs that all employees and visitors must follow.
For diseases already established on a farm, a variety of types of live and killed vaccines are available that should be included in a logically designed vaccination program that will provide the needed immunities without overly stressing the growing flocks.
When designing vaccination programs, consideration should be given to what diseases need to be included, maternal antibody levels in the chicks, what vaccines are available, and preference or ability to administer live versus killed vaccines, and spray versus drinking water live vaccines.
Some respiratory disease vaccines are only available in a live form (LT), some only in a killed form (Coryza), but most are available in either form. In some cases, we only need to use one form, live or killed, such as for MG. In other cases (Newcastle disease and Bronchitis), we typically use live vaccination first to stimulate early protection and to “prime” the immune system against that antigen. Then we follow the live vaccinations with a final killed vaccine injection which boosts the titer higher and provides long lasting protection.
Designing the vaccination program is just the first step; then proper vaccination techniques need to be used that will uniformly immunize all birds in the flock.
Live respiratory vaccinations are probably best applied individually by eyedrop application, but often the mass application methods of spray or drinking water are used to save on labour expense. The challenge for these methods in large commercial flocks is getting all birds uniformly exposed to the vaccine.
For drinking water application, it is best for the birds to be thirsty so they will consume the vaccine-treated water within 1 - 2 hours. This is best accomplished when the lights first come on in the morning. Most respiratory viruses will die off and be ineffective after two hours. It is important to use enough water so the vaccine will be consumed by essentially all birds in the house, but not so much that it won’t be consumed and will just sit and become inactive in the water system. This requires careful planning and practice before actually conducting a vaccination.
The spray route may be easier to administer, but again, the proper volume needs to be used so that vaccine can be applied uniformly to all birds in the house. The lack of a uniform response to some vaccine viruses, like LT, results in the vaccine spreading between individuals and reverting to virulence as it does so. In this manner, an “outbreak” of LT can be caused by poor vaccination technique.
All live vaccines are susceptible to inactivation by sanitizing chemicals that may be in the water. Vaccine stabilizers such as milk based products or newer second generation products should be used to neutralize any chlorine in the water.
Subsequent serology testing will verify the immune status of the flock and identify areas of deficiency in the vaccine application process. The antibody response will generally take 2 - 4 weeks to develop after vaccination. Blood samples should be drawn from a representative number of birds in the flock.
The serum is then separated and can be frozen for storage or shipping to a laboratory. Common laboratory techniques include the serum plate agglutination test or ELISA for MG, and ELISA for Newcastle and bronchitis, and perhaps LT, AI, and pneumovirus.
Vaccination generally induces a detectable antibody response, although not all vaccines do, so it is important to evaluate serology results with the vaccination program in mind. With some experience interpreting serology titres, one can determine if there has been a proper level of antibody response to the vaccinations administered, and whether the response is uniform among the individual birds.
Image 3 Dupont BAX Salmonella PCR
Field virus disease exposure will either turn a flock antibody positive for that disease (if not previously vaccinated) or may significantly raise the level of antibody titer if the flock was previously vaccinated. As such, serology may strongly indicate the presence of a disease, but other diagnostic techniques may need to be used as a confirmation as mentioned earlier. Low individuals on histograms likely indicate birds that missed the killed injection.
^ Top page
.
VECTOR VACCINES
Some new genetically engineered vector vaccines are now available that have the potential to provide immunity without the risk of respiratory reactions typical with traditional modified live vaccines. Vector vaccines utilize a carrier virus which in the currently licensed products is a common vaccine virus made for fowl pox or the HVT vaccine for Marek’s disease.
These viruses are genetically manipulated to carry some important genes from other disease viruses that will produce immunogenic proteins from those viruses. So, as the pox or HVT virus replicates in the bird, this foreign disease protein is also produced and stimulates immunity against that virus without the stress of reacting to the whole infectious virus.
Pox vectors have been produced for protection against infectious laryngotracheitis, Newcastle disease, influenza, and MG. HVT vectors are available for infectious laryngotracheitis, IBD, and Newcastle disease.
When vector vaccines are used, in order to get the best response to the foreign gene component, it is important to get a good strong response to the vector virus so that it can fully express the immunogenic genes of the secondary virus.
For this reason, it is critical that the vector product is the bird’s first exposure to that virus. For example, if a pox vector is to be used at 8 - 12 weeks of age, the flock should receive no earlier regular pox vaccination at hatch or any time prior to the vector vaccination. Likewise, the HVT vector products should not be mixed together with other regular HVT or vector HVT vaccines.
Perhaps in the future we will have vector vaccines that express more than one foreign disease protein, but at present, we can only select a single HVT vector and a single pox vector from the available products.
Management of the respiratory diseases involves many considerations, but is a critical aspect to maintaining the health and productivity of layers around the world.
^ Top page
This article appeared in International Poultry Production, Volume 20 Number 4 (2012). ©Copyright 2012, All Rights Reserved.

 Corporate Website
Corporate Website
 Africa
Africa
 Argentina
Argentina
 Asia
Asia
 Australia
Australia
 Belgium
Belgium
 Brazil
Brazil
 Bulgaria
Bulgaria
 Canada (EN)
Canada (EN)
 Chile
Chile
 China
China
 Colombia
Colombia
 Denmark
Denmark
 Egypt
Egypt
 France
France
 Germany
Germany
 Greece
Greece
 Hungary
Hungary
 Indonesia
Indonesia
 Italia
Italia
 India
India
 Japan
Japan
 Korea
Korea
 Malaysia
Malaysia
 Mexico
Mexico
 Middle East
Middle East
 Netherlands
Netherlands
 Peru
Peru
 Philippines
Philippines
 Poland
Poland
 Portugal
Portugal
 Romania
Romania
 Russia
Russia
 South Africa
South Africa
 Spain
Spain
 Sweden
Sweden
 Thailand
Thailand
 Tunisia
Tunisia
 Turkey
Turkey
 Ukraine
Ukraine
 United Kingdom
United Kingdom
 USA
USA
 Vietnam
Vietnam