...
-->Introduction
-->Maternal Antibody Transfer from the Hen to the Progeny
-->Newcastle Disease Virus (NDV)
-->Infectious Bronchitis Virus (IBV)
-->Infectious Laryngotracheitis (ILT)
...
PASSIVE IMMUNITY: PART 1
By Dr. Roberto SOARES, DVM, MSc., ACPV – Ceva Santé Animale – Libourne, France
.
Introduction
Maternal Derived Antibodies (MDA) or also known as Passive Immunity are the naturally transfer of immunoglobulins from one individual to another. In birds, maternal antibodies are passed from hyper-immunized or naturally infected breeder hens to the progeny through the egg. This Passive Immunity has relatively short duration, commonly 1-2 weeks and generally less than 4 weeks and its function is to protect young chicks during a period (first few weeks) when their immune system is not fully developed to proper react to an early challenge.
^ Top page
.
Maternal Antibody Transfer from the Hen to the Progeny
The transfer of antibodies to the embryo occurs in two steps. First, the antibodies are deposited in the egg yolk and albumin (egg white) and afterwards it is transferred to the embryo.
a. MDA Transfer from the Hen to the Egg
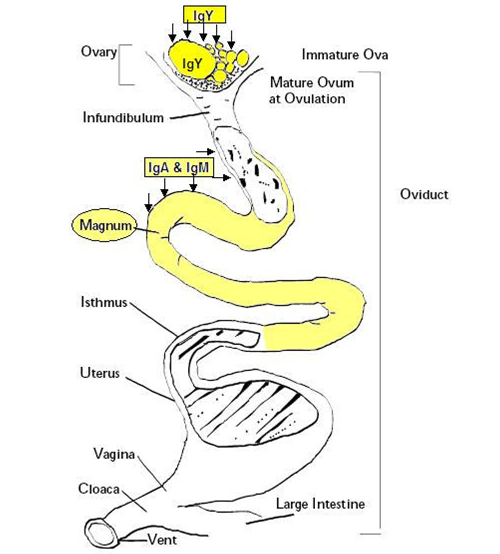
The hens transfer MDA to the egg by depositing the antibodies [IgY, IgA and IgM)] in the egg yolk and albumin. As chicken IgG molecule is longer than mammalian IgG, the chicken IgG is referred by some authors as IgY. However, avian IgG (or IgY) is functionally homologous to mammalian IgG (Sharma, 1997). The pathway of depositing immunoglobulin (Ig) in the egg differs between immunoglobulins.
The IgY is the most predominant Ig isotype in the egg yolk. This Ig is secreted by the chicken ovarian into the developing ova (egg yolk) in different stages.
The passage of IgY into the ova is regulated by the follicular epithelium which goes through morphologic changes as the ova grow. This epithelium becomes flatter and thinner in larger ovum allowing the passage of a large amount of IgY. The transfer of IgY through the ovarian follicular epithelium reaches its maximum 3 to 4 days prior ovulation and starts to decrease due to the development of vitelline membrane between the ovum and the follicular epithelium of ovary in preparation for ovulation.
Therefore, as a single hen has several ovas in different stage of development, the amount of IgY transferred to each one is not the same.
The IgA and IgM are mainly found in the albumen (Rose et al., 1974) and they are transferred to the albumen as a result of mucosal secretion in the oviduct more specifically in the Magnum.
Image 1 MDA Transfer from the Hen to the Egg
.
b. MDA Transfer from the Egg to Embryo
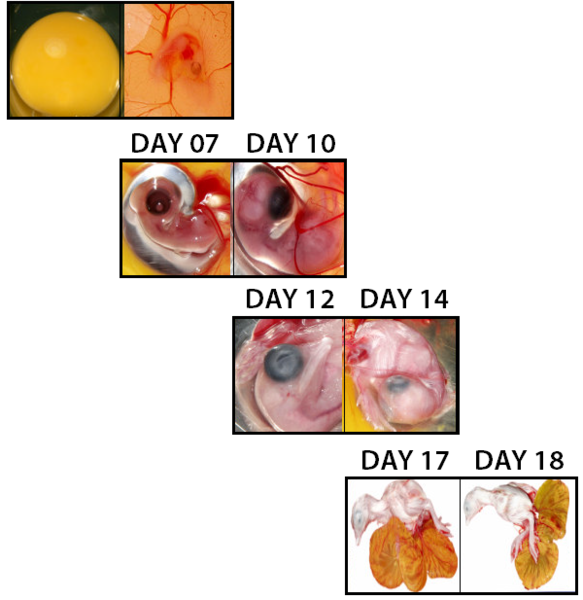
The IgY is transferred from the egg yolk to the offspring via the embryonic circulation. The transfer starts from day 7 of embryonic development and reaches its maximum rate 3 to 4 days before hatch.
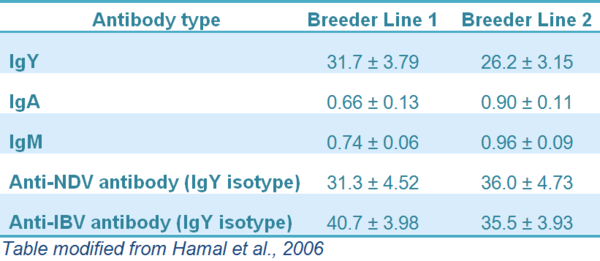
The amount of IgY transferred to the egg yolk and from the egg yolk to the embryo has been reported to be proportional to maternal serum IgY concentrations. In a work done by Hamal et al. (2006), they found that 27 to 30% of hens IgY is transferred to the progeny (Table 1).
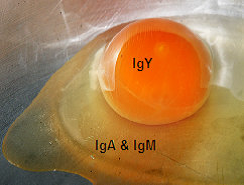
Image 2 The presence of IgY, IgA & IgM in egg
Image 3 Embryonic development
IgA and IgM are transferred to the embryo by absorption of the albumen by embryonic gut and may have its major function in the newly hatched chick as a protective Ig in the alimentary tract or as an additional source of protein.
The amount of IgA and IgM transferred to the progeny is less than 1% of the concentration of these Ig in the hens’ plasma (Table 1). Besides of low percentage transferred, IgM is the first Ig isotype to be synthesized by the newly hatched chick followed by the IgA and IgY.
^ Top page
.
MDA Anti-Specific Agents
The efficacy of MDA in protecting young chicks is variable and depending on several factors such as MDA level and the agent involved. In the Part 1 of this review we will focus in MDA of three agents that cause respiratory diseases: Newcastle Disease virus (NDV), Infectious Bronchitis virus (IBV) and Infectious Laryngotracheitis virus (ILTV).
However, it is important to stress the fact that it is widely accepted practice to vaccinate poultry against various diseases including Newcastle Disease and Infectious Bronchitis by coarse-spray at the hatchery, even though the birds may possess maternally derived antibody. The rationale for this practice is that the vaccine is beneficial in inducing a local immune response even though maternal antibodies generally interfere with the systemic response (as measured by serologic antibody).
Table 1 Progeny plasma Ig concentration as a percentage of the maternal plasma Ig concentration
.
Newcastle Disease Virus (NDV)
The anti-NDV antibody derived from the hen provides protection for young chicks. Hamal el al (2006) found that the level of NDV-specific antibodies transferred from the hen to the progeny range between 27 and 40% and it is directly related to titres in the hen (Table 1). IgY is also found in the tear of day-old chick at the rate of 1:5 of the serum level (Russell, 1992).
This MDA anti-ND starts to be catabolized as soon the chick hatch. According to Allan et al. (1978) every 4.5 days, twofold of maternally derived HI titer is catabolized by the chicks.
The protection provided by the MDA anti-ND also interferes with the systemic replication of vaccine strains if applied in the presence of high MDA. Therefore, the objective of day-old vaccination with live ND vaccine, as aforementioned, is to efficiently prime the birds, stimulate the local immunity (Cell Mediated Immunity) in the upper respiratory tract and induce early protection in chicks with low MDA.
Oil-emulsion inactivated vaccines have been successfully used in day-old chicks with maternal immunity in the prevention of ND (Alexander and Jones, 2001). The major advantages of those inactivated vaccines are the very low level of adverse reactions in vaccinated birds and extremely high levels of protective antibodies of long duration that can be achieved (Alexander and Jones, 2003).
Moreover, these inactivated oil-emulsion vaccines are not as adversely affected by maternal immunity as live vaccines (Box et al., 1976) because the oil adjuvant acts as stimulus of defense mechanism and disperse antigen slowly. In these circumstances, there is a progressive stimulation of the active immunity while the passive immunity declines and the immune system reaches full competence (Bennejean et al., 1978; Box et al., 1976; Warden et al., 1975.)
^ Top page
.
Infectious Bronchitis Virus (IBV)
Maternally-derived antibodies for IB virus differ from flock to flock and this is mainly caused by factors like the vaccine strains used, vaccination programs, quality of vaccine application, production systems and bird line. The percentage IgY-anti IBV transferred from the hen to the progeny range between 31 and 41% (Table 1).
MDA for IBV has been demonstrated to be protective. Mondal and Naqi (2001) observed that chicks with high MDA titers anti-IBV had more than 95% protection against IBV challenged at one day of age, however the MDA for IBV seems to decline fast and in the same paper those authors found that the protection at seven days were less than 30%. This is in agreement with what was found by Hamal et al (2006) where they observed that MDA decreased substantially at day 7, and were no longer detected at day 14.
The strong protection observed by the above authors is concluded to be due to the high level of local protection. Because of this, IBV vaccination of maternally immune one-day-old commercial chicks is routinely performed, regardless of reduced humoral immune response in MDA positive chicks.
Talebi et al (2005) found that the MDA anti-IBV of unvaccinated chicks decline slightly faster (half-life of 5 days) than MDA of chicks vaccinated at day one with IB H120 by spray, eye-drop and drinking water methods (half-life of 6 days).
In countries where IB variants strains are present, breeders should to be vaccinated with these strains in order to produce specific MDA.
^ Top page
.
Infectious Laryngotracheitis (ILT)
Offspring of breeders vaccinated to ILT receives MDA via egg. However, this maternal antibody does not confer protection against infection or interfere with vaccination (Fahey et al., 1983).
Davison et al (1989), assessing the protection provide by ILT maternal antibodies during the first 4 weeks of chick’s live, found that chicks from all ages tested (1, 7, 14, 21 and 28 days) were susceptible to infection.
Due to the fact that ILTV infections are usually limited to the upper respiratory tract and viremia is rarely observed, maternal antibodies and secretory immunoglobulins do not correlate well with protection. Protection to ILT virus seems to be mediated primarily by the cellular immune response. Therefore, these points must be taken into consideration in the development of a vaccination strategy against ILT.
^ Top page
.
REFERENCES
Alexander, D.J and Jones, R.C. 2001. Newcastle Disease. In: F.T.W Jordan (Ed.) Poultry Diseases, 5th Edition, WB Sanders.
Alexander, D.J and Jones, R.C. 2003. Newcastle Disease, Other Avian Paramyxovirus, and Pneumovirus Infections. In: Y.M. Saif (Ed.) Diseases of Poultry, 11th Edition, p. 63-92. Iowa State Press.
Allan, W. H., J. E. Lancaster, and B. Toth. 1978. Newcastle disease vaccines—Their production and use. FAO Animal Production and Health Series No. 10. FAO: Rome, Italy.
Bennejean, G., Guittet, M., Picault, J.P., Bouquet, J.F., Devaux, B., Gaudry, D. Moreau, Y. 1978. Vaccination of day-old chicks against Newcastle Disease using inactivated oil adjuvant vaccine and/or live vaccine. Avian Pathology, v. 7, n.1, p. 15-27.
Box, P.G., Furminger, I.G.S., Robertson, W.W, Warden, D. 1976. The effect of Marek’s Disease vaccination on immunity of day-old chicks against Newcastle Disease, using B1 and oil emulsion vaccine. Avian Pathology, v.5, p. 299-305.
Cardoso, W.M. , Gomes, L.P. , Romão, J.M.,. Salles, R.P.R., Teixeira, R.S.C, Sobral, M.H.N.R., Câmara, S.R., Oliveira, W.F. 2006. Antibodies specific to infectious bronchitis in broilers in Ceará state, Brazil. Arq. Bras. Med. Vet. Zootec., v.58, n.3, p.327-332
Davison, S., Smith, G., Eckroade, R.J. 1989. Laryngotracheitis in Chickens: Infectious Studies and the Efficacy of Tissue-Culture vaccine in chicks less than four weeks Age. Avian Diseases 33: 24-29.
Fahey KJ, Bagust TJ, York JJ. 1983. Laryngotracheitis herpesvirus infection in the chicken:The role of humoral antibody in immunity to a graded challenge infection. Avian Pathology: 12:505-514.
Hamal, K.R., Burgess, S.C., Pevzner, I.Y. and Erf, G.F. 2006. Maternal Antibody Transfer from Dams to Their Egg Yolks, Egg Whites, and Chicks in Meat Lines of Chickens. Poultry Science 85:1364–1372
Heller, E. D., D. B. Nathan, and M. Perek. 1977. The transfer of Newcastle serum antibody from the laying hen to the egg and chick. Res Vet Sci 22:376—379.
Hidalgo, H. 2003. Infectious Laryngotracheitis: A Review. Brazilian Journal of Poultry Science : v.5 , n.3, p. 157 – 168
Hussell, P.H. 1993. Newcastle Disease Virus: virus replication in the Harderian gland stimulates lacrimal IgA, the yolk sac provides early lacrimal IgG. Vet. Immunol. Immunopathol., 37: 151-163.
Kramer, T. T., and H. C. Cho. 1970. Transfer of immunoglobulins and antibodies in the hens egg. Immunology 19:157–167.
Leslie, G. A., and W. L. Clem. 1969. Phylogeny of immunoglobulin structure and function. 3. Immunoglobulins of the chicken. J. Exp. Med. 130:1337–1352.
Mondal, S.P.; Naqi, S.A. 2001. Maternal antibody to infectious bronchitis virus: its role in protection against infection and development of active immunity to vaccine. Vet. Immunol. Immunopathol., v.79, p.31-40.
Patterson, R. , Youngner, J.S., Weigle, W.O. and Dixon, F. J. 1962. The Metabolism of Serum Proteins in the Hen and Chick and Secretion of Serum Proteins by the Ovary of the Hen. The Journal of General Physiology 45:501-513
Sharma, J.M. 1997. The structure and function of the avian immune system. Acta Veterinaria Hungarica, v. 45, n. 3, p.229-238.
Talebi,A., Pourbakhsh, S.A., and Dorostkar, K. 2005. Effects of Vaccination Routes Against IB on Performance and Immune Responses of Broiler Chickens. Intern. J. of Poultry Science 4 (10): 795-798.
Rose, M. E., E. Orlans, and N. Buttress. 1974. Immunoglobulin classes in the hen’s egg: Their segregation in yolk and white. Eur. J. Immunol. 4:521–523.
Warden, D. Furminger, I.G.S., Robertson, W.W. 1975. Immunizing Chicks against Newcastle Disease by Concurrent Inactivated Oilemulsion and Live B1 Vaccines. Veterinary Record, v.18, p. 65-66.
If you need to download this article, please do not hesitate to contact us!

 Corporate Website
Corporate Website
 Africa
Africa
 Argentina
Argentina
 Asia
Asia
 Australia
Australia
 Belgium
Belgium
 Brazil
Brazil
 Bulgaria
Bulgaria
 Canada (EN)
Canada (EN)
 Chile
Chile
 China
China
 Colombia
Colombia
 Denmark
Denmark
 Egypt
Egypt
 France
France
 Germany
Germany
 Greece
Greece
 Hungary
Hungary
 Indonesia
Indonesia
 Italia
Italia
 India
India
 Japan
Japan
 Korea
Korea
 Malaysia
Malaysia
 Mexico
Mexico
 Middle East
Middle East
 Netherlands
Netherlands
 Peru
Peru
 Philippines
Philippines
 Poland
Poland
 Portugal
Portugal
 Romania
Romania
 Russia
Russia
 South Africa
South Africa
 Spain
Spain
 Sweden
Sweden
 Thailand
Thailand
 Tunisia
Tunisia
 Turkey
Turkey
 Ukraine
Ukraine
 United Kingdom
United Kingdom
 USA
USA
 Vietnam
Vietnam